Why use alum when Ferrate is proven safer and better?
Disclosure: Some of the material presented herein was extracted from the Everglades Consolidated Report. Annual Research Report, South Florida Water Management District (2002), unless otherwise noted. This work was performed under the direction of the South Florida Water Management District (SFWMD) since 1996 at several sites in Central Florida. The focus of the work was to compare different “advanced” treatment technologies for the removal of phosphorus from stormwater runoff (bench scale flows between 2 – 20 GPM).
Overall Summary of 25 Years of Research
Florida continues to struggle with controlling nutrient inputs to our fresh and marine waters, and HABs are increasing in periodicity and intensity. Scientists have been studying the excessive buildup of algae in Florida waters for years, and they have progressed to the point that numerical estimates have now been made about nutrient limitations required to prevent excessive algal growth.
Because Florida’s progress in this endeavor has long been handicapped by hanging on to old beliefs that simple biological systems (grass, wetlands, oysters etc.) could remove enough nutrients to balance the massive influx from domestic waste and agricultural runoff, very little work has been done to develop effective, inexpensive, and safe chemical systems that can actually solve the problem.
Unfortunately, the only chemical systems tested by the State at the pilot scale were the usual cast of characters; Calcium (carbonate or hydroxide), Aluminum (hydroxide, chloride or sulfate) & Iron (ferric). These characters can only precipitate phosphorus from the water at low efficiency, and their effectiveness is highly dependent on background environmental chemistry.
Summarizing all the results to date; it can be shown that these “advanced technologies” are not effective enough to reliably remove the mass of N & P required to control excessive algal growth.
Across the entire data base, P removal efficiencies are only between 35% – 60%. Total P removals need to be > 95% to be an effective treatment technology. In addition, only free ortho-phosphate was reliably removed by the chemicals tested. Other common forms of phosphorus, e.g. particulate organic & inorganic bound phosphorus forms, were not easily removed.
Advanced Chemical Treatment Technologies
Iron and aluminum compounds have been utilized for more than 100 years to coagulate suspended material from water; mostly utilized for drinking water treatment. It was also discovered that these compounds could co-precipitate phosphorus from water, but their use for this treatment has been limited. The choice between aluminum or iron was usually determined by the cost, as certain regions have iron supplies readily available, and others have aluminum. However, there is a major difference between these two metals in terms of toxicity. Iron is the most common element on our planet and has no toxicity to humans; in fact most humans are iron deficient. Aluminum on the other hand is extremely toxic and has been implicated in chronic illnesses of humans such as Alzheimer’s disease. This toxicity has limited the use of aluminum salts in water treatment in some areas, especially concerns about the discharge of aluminum sludge to the environment. ALUMINUM IS TOXIC!
Over 68,000 Scientific Publications Addressing Aluminum Sludge Toxiciy (Google)

The inherent toxicity associated with aluminum sludge generated from water or wastewater treatment applications has not been a publisized issue to date, as its use has been limited. If Florida begins utilizing large volumes of aluminum for removal of phosphorus, the massive amount of sludge disposal is going to become an environmental issue.
That leaves iron as the only safe treatment for removal of phosphorus on a large scale, according to previous studies. However, the limitations of iron as utilized in previous studies (ferric iron) in terms of nutrient removal efficiency remains an issue.
It’s Time For A Truly New,
Innovative Technology
Innovative Technology
Ferrate
A truly new and innovative technology, now commercially available, that can address all of these issues is iron-six Ferrate (FeO42-). This is a form of iron that has been super charged to a high valance which gives it oxidizing power as well as coagulation capability. Because of this oxidizing capacity, Ferrate is able to release phosphorus in all of its forms into a form that can be precipitated on the iron hydroxide. This means that Ferrate is able to remove total phosphorus to levels that are well below anything achievable by ferric iron. Ferrate also has the redox potential to oxidize reduced nitrogen (ammonia) directly to N2 gas. Therefore, both total P and total N are removed concurrently with one treatment. NO OTHER TREATMENT SYSTEM ON THE PLANET CAN DO THIS.
What is in Storm Water Runoff?
Storm water runoff contains all kinds of environmental contaminants, including oil, pesticides, heavy metals, and nutrients (N & P). While nutrients are the focus of this discussion, it is important to understand that many different contaminants and toxins are also present. Because ferrate is both an oxidant and coagulant, it can precipitate or destroy all forms of the contaminants in storm water runoff.
Where is Ferrate Working?
Ferrate treatment has been quietly working for over a year on removing both N & P from the muck dredge water on the Indian River Lagoon in eastern Florida. Below is a photo of the ferrate treatment system operating on the IRL site. It operates six days a week, treating 4,000 GPM. In the past eight months alone, ferrate has removed over 30 TONS! Of nitrogen and over 2 TONS! of phosphorus.

Cost and Efficiency
Ferrate systems are affordable, and given the efficiency of near total removal of both N & P, as well as other contaminants noted above, it is unlikely that any other treatment in the C-43 Basin could come close to its low cost when measuring pounds, or tons, of contaminate removal.
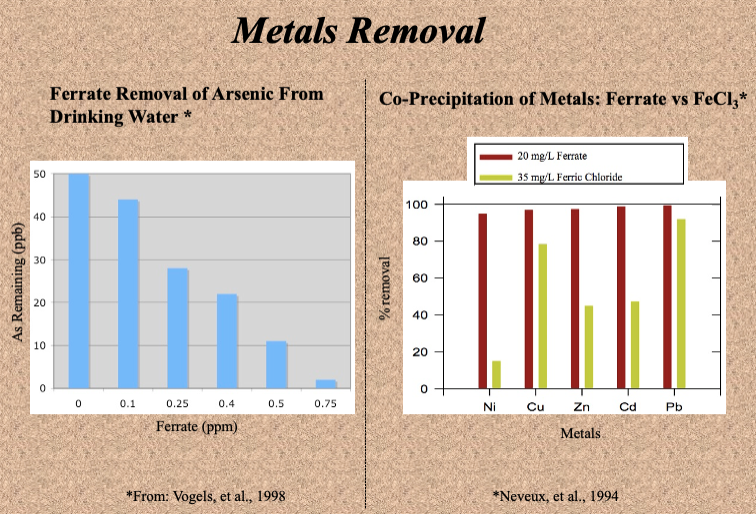
Comparison of Ferrate to Ferric Iron Treatment

Ferrate Sludge Returned to Agricultural Lands


Leave a Reply
You must belogged in to post a comment.